HOME>Experiments>For Kitchen>EH007 Citric Acid Lemonade
The sourness of lemon juice comes from citric acid. By measuring the amount of baking soda used for neutralization, you can determine the amount of citric acid present in the lemon juice.
Will lemonade made with lemon juice taste the same as lemonade made with citric acid?
The sourness of lemonade puts a strain on the digestive system (especially on an empty stomach). Be careful not to drink too much.
Requires
equipments
・deep cup or glass
・measuring cup
・scale
reagents
・baking soda
・citric acid
・lemon juice
Methods
1.

Pour 2 tablespoons of lemon juice into a measuring cup, then add water until the total volume reaches 100 mL. Stir well, then transfer it to a glass.
2.

Add 1 g (or 0.5 g) of baking soda at a time, and stop when no more bubbles are produced after adding the baking soda. Finally, exclude the last amount of baking soda added and note down the total amount of baking soda you have used up until that point.
3.

Prepare five empty cups and add 1 g, 2 g, 3 g, 4 g, and 5 g of citric acid to each cup, respectively. Then, add 100 mL of water to each cup and dissolve the citric acid.
4.

Perform the same procedure as in step 2 for each of the citric acid solutions.
5.
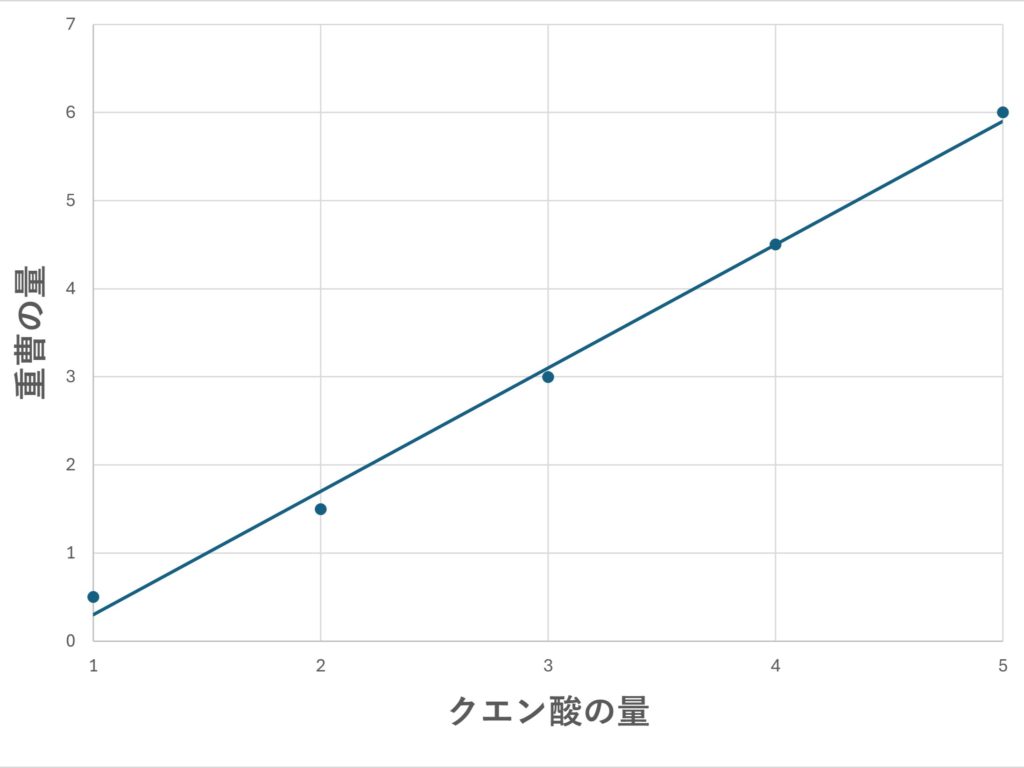
Create a graph with the weight of citric acid on the horizontal axis and the amount of baking soda (noted in step 4) on the vertical axis. Afterward, use the graph to find the corresponding amount of citric acid for the amount of baking soda you recorded in step 2.
6.

Make the lemon water again as in step 1, and in a separate cup, dissolve the amount of citric acid determined in step 5 into 100 mL of water. Taste both, and compare the sourness and flavor. (You can also add an equal amount of sugar or simple syrup to both if desired.)
Clearing
Dispose of all the liquids from the experiment down the drain.
Principle
In experiment methods ② and ④, continue repeating the process of adding powder → stirring and waiting for the bubbles to subside → adding more powder, and so on. Stop when no more bubbles are produced after adding the powder.
Baking soda reacts with the acidic components (H+) in lemon juice, releasing carbon dioxide. The bubbles that form are a result of this reaction.
NaHCO3+H+→Na++CO2+H2O
The “pH,” which measures the strength of acidity or alkalinity, changes based on the amount of H+ ions present in water. It can be a bit complicated, but the higher the concentration of H+, the closer the pH is to 0 (strongly acidic), and the lower the concentration of H+, the closer the pH is to 14 (strongly alkaline). Lemons typically have a pH of 2, which means they are strongly acidic.
As shown in the earlier chemical reaction, when baking soda consumes H+ ions to produce carbon dioxide, the amount of H+ decreases. As a result, the pH increases, meaning the solution becomes more alkaline.
In addition, the citric acid present in lemon juice releases three H+ ions per molecule. This means that when baking soda reacts with one molecule of citric acid, it produces three carbon dioxide molecules. *However, it is important to note that the three H+ ions are not consumed all at once.
When baking soda is added to lemon water or citric acid water, at some point, the bubbles will stop. This indicates that the neutralization process is complete, and the pH will be close to neutral.
There are several other methods to measure the strength of acidity besides this one. Let’s explore some more accurate methods.

