HOME>Experiments>For Kitchen>EH013 Slime
Let’s make some slime that everyone loves!
Requires
equipments
・disposable splittable chopsticks
reagents
・borax
・glue (containing PVA)
Methods
1.

Add 4 g of borax and 60 mL of water to a suitable container. Then, dissolve the borax thoroughly.
2.

In another container, add 200 mL of laundry starch and 100 mL of water. Mix them together using chopsticks.
3.

Add the liquid from step 1 to the liquid from step 2 and mix them together using the chopsticks.
4.

After mixing for a while, large clumps will form. Take it in your hands and knead it well.
Clearing
After playing with the slime, place it in a plastic bag and dispose of it as burnable waste. The containers used can be washed as usual.
Principle
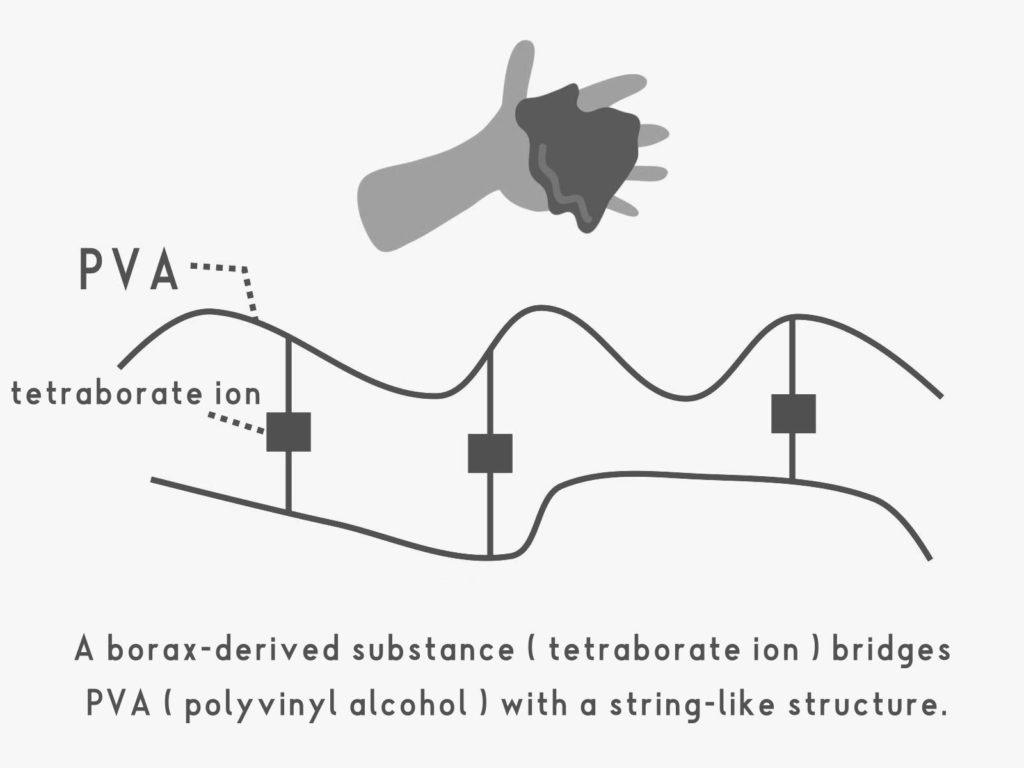
PVA (polyvinyl alcohol) is a substance where multiple vinyl alcohol units are linked together, forming a long, string-like structure, and is a type of “polymer” (a macromolecule). Other types of polymers include PET (polyethylene terephthalate), PS (polystyrene), rubber, cellulose, and many more.
When borax is dissolved in water, tetrahydroborate ions are formed. These tetrahydroborate ions act like bridges, connecting the PVA molecules. As a result, the string-like PVA molecules become tangled, forming clumps.
A similar mechanism can be observed in natural rubber, which is made by mixing sulfur with latex. The sulfur molecules act as bridges, connecting the string-like molecules in the latex.

