HOME>Experiments>For Kitchen>EH012 Blue Bottle reaction
Methylene blue changes its color or restores it due to the interaction with glucose and dissolved oxygen in the liquid.
Ammonia water should be handled in a well-ventilated area. Be sure not to use the undiluted solution for experiments; always dilute it with water as instructed in the experimental procedure.
Requires
equipments
・empty plastic bottle
・sticky notes or masking tape
・measuring cup
reagents
・methylene blue for aquarium
・ammonia water (10 w/v%)
・gum syrup
Methods
1.

Mix 80 mL of water and 20 mL of ammonia water in a measuring cup.
2.

Pour the diluted ammonia water into an empty plastic bottle. Add 2–3 drops of aquarium methylene blue and the equivalent of two servings of syrup.
3.

Gently shake the bottle until the entire liquid turns blue, then leave it to stand for a while. Over time, the blue color will fade.
4.

Shake the bottle vigorously. After shaking for a while, the blue color will gradually return.
5.

What happens if you stop shaking and wait for a while?
Clearing
Dispose of the liquid in the bottle by pouring it down the drain while running tap water to dilute it. Make sure to keep the ventilation fan on for a while during the process.
Principle
The blue methylene blue is converted into a colorless substance called leucomethylene blue by the glucose contained in the syrup. Shaking the bottle dissolves oxygen from the air into the liquid, which reacts with the leucomethylene blue, turning it back into methylene blue.
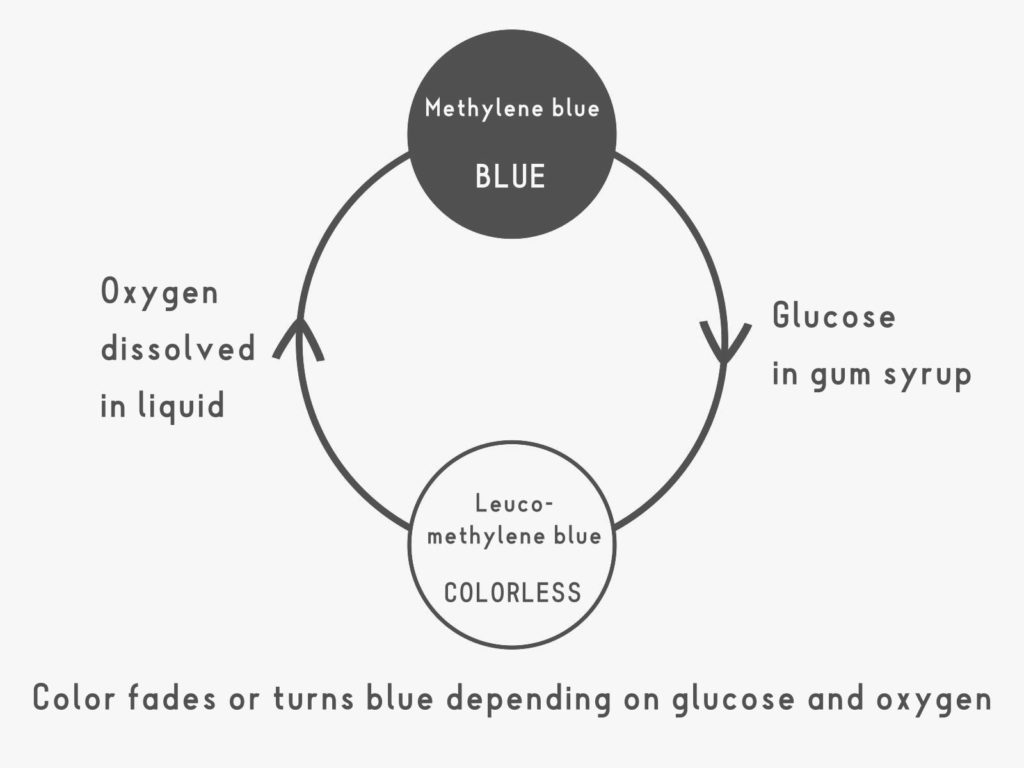
Over time, glucose again converts methylene blue into leucomethylene blue, but the speed of this reaction (reaction rate) depends on factors such as the amount of glucose and the temperature of the liquid. Try changing the experimental conditions to see how these factors affect the time it takes for the color to change.

