HOME>Experiments>For Lab>EL003 Nylon 66 Capsules
Everyone is familiar with nylon 66 from fiber spinning experiments, but actually, the same material can be used to create capsules with semi-permeable properties.
After the experiment, make sure to dispose of the waste liquid according to the rules of the laboratory.
When it comes to nylon 66, the well-known experiment where a membrane is formed at the interface of two different solutions and then lifted with tweezers is famous. However, there is also an experiment that utilizes the semipermeability of this membrane, which I would like to introduce.
Requires
equipments
・100 mL beaker×2
・petri dish
・dropper
reagents
・adipoyl dichloride
・ hexamethylene diamine
・hexane
・sodium bicarbonate
・saturated saline solution
Preparation
Hexamethylenediamine is a solid at room temperature, but since its melting point is 42°C, it can be dissolved in hot water along with the reagent bottle and then measured using a pipette. Here, I will introduce a method to use it in its solid form.
Methods
1.
Add 30 ml of hexane to a beaker and dissolve 1.0 g of adipoyl chloride in it. This will be referred to as “Solution A.” Pour Solution A into a petri dish, filling it up to a height of 3 to 5 mm from the top of the walls.
2.
In a separate beaker, add 30 mL of pure water, dissolve 0.5 g of sodium bicarbonate, and 1.0 g of hexamethylenediamine. This will be referred to as “Solution B.”
*since sodium bicarbonate is used to neutralize adipoyl chloride, it can be substituted with other compounds such as sodium carbonate.
3.
Take Solution B with a dropper and add it drop by drop to Solution A in the petri dish.
4.

Remove Solution A from the petri dish and add saturated saline solution. After waiting for a while, you will notice that the capsule shrinks.
5.
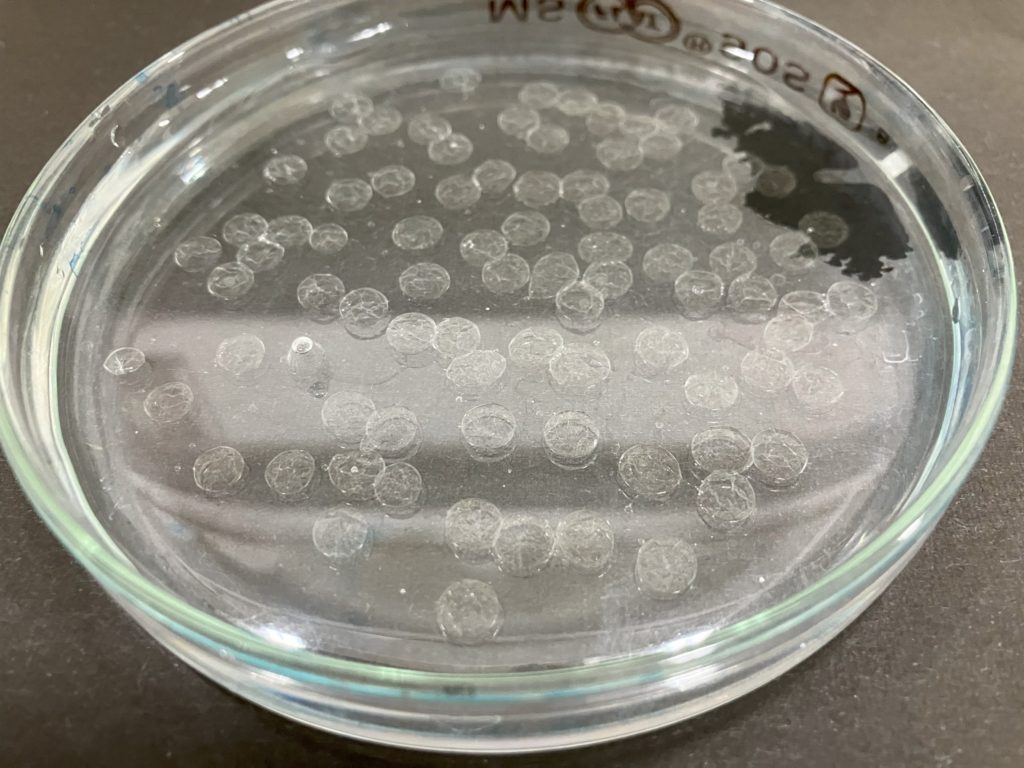
After removing the saturated saline solution, add pure water. You will observe that the shriveled capsule begins to swell.
Principle
It is recommended to observe the capsule by following the procedure in steps 4 and 5, using saturated saline solution and pure water in that order. If this is done in reverse, the capsule may break, similar to how red blood cells undergo hemolysis in water, due to the osmotic pressure differences.
reference
・関 隆広, 岡畑 恵雄, 海老原 真純, “ナイロンでカプセルをつくる : 界面重合法によるナイロンカプセル膜の調製(デモ実験虎の巻)” 化学教育, 34(6), 1986, p.492-493
・平山美樹, “高分子らしさを体感する実験” 化学と教育, 68(9), 2020, p.376-379

